This fact box is is one of three fact boxes designed to help you weigh the benefits and side effects of the MMR combination vaccination against mumps, measles and rubella in young children. The focus of this fact box is on mumps. The information and figures do not offer a definitive assessment. They are based on the best current scientific evidence available.
The fact box was produced by the Harding Center for Risk Literacy.
Mumps is a contagious infectious disease of the salivary glands caused by the mumps virus. It is usually transmitted through direct contact with saliva (e.g. when kissing), but also through droplets of saliva that are transmitted when coughing and speaking. Less frequently, the mumps virus can be transmitted via surfaces and objects contaminated with saliva (e.g. door handles, drinking bottles) [1, 2].
In the first phase of the disease, symptoms similar to a those of a cold often appear, such as fever, cough, and headache. Later, there is inflammation of the parotid glands, either on one or both sides of the face. This leads to the painful swollen cheeks typical of mumps. The salivary gland under the tongue can also become inflamed and swollen, as can the lymph nodes in the neck. The swelling subsides after three to eight days [1, 2].
A mumps infection is usually harmless in childhood, but it can also cause inflammation of various glands and organs and have long-term consequences. These are more frequent in adults who contract mumps than in children [1, 2]:
- In about 15 to 30 out of every 100 men,
- inflammation of the testicles occurs, which can permanently reduce fertility.
- Breast inflammation occurs in about 30 out of every 100 women, and inflammation of the ovaries in about 5 out of every 100 women.
- Inflammation of the brain can occur in both sexes, in about 1 in every 100 people.
- Less frequently, inflammation of the pancreas and the auditory nerves can occur. Inflammation of the auditory nerves can lead to loss of hearing.
Since the introduction of the vaccine against mumps, only about 1 in every 100,000 people in Germany contracts the disease. Most of those who get mumps are adolescents and younger adults [2].
The mumps-containing vaccine is given twice to infants as part of a triple combination vaccination also for measles and rubella (MMR) or as part of a quadruple combination vaccination also for measles, rubella, and chickenpox (MMRV). After a double vaccination, a lifelong immunity to mumps is generally assumed so that, according to the current state of sciencethe vaccination protection does not need to be refreshed later. In most cases, complete vaccination protection exists three to four weeks after the second vaccine dose [2].
The Standing Committee on Vaccination (STIKO) of the Robert Koch Institute (RKI) recommends administering the first MMR(V) vaccine dose as part of basic immunization. This takes place at the age of 11-14 months. The second MMR(V) vaccine dose can be given at the earliest four weeks after the first dose, and the vaccination should thus be completed by the age of 15-23 months. The second vaccine dose is considered a precautionary measure in case sufficient immunity was not developed during the first vaccination [2].
Furthermore, all adults born after 1970 who were not vaccinated in childhood, who were vaccinated only once, or whose vaccination status is unclear may consider a single MMR(V) combination vaccination [2].
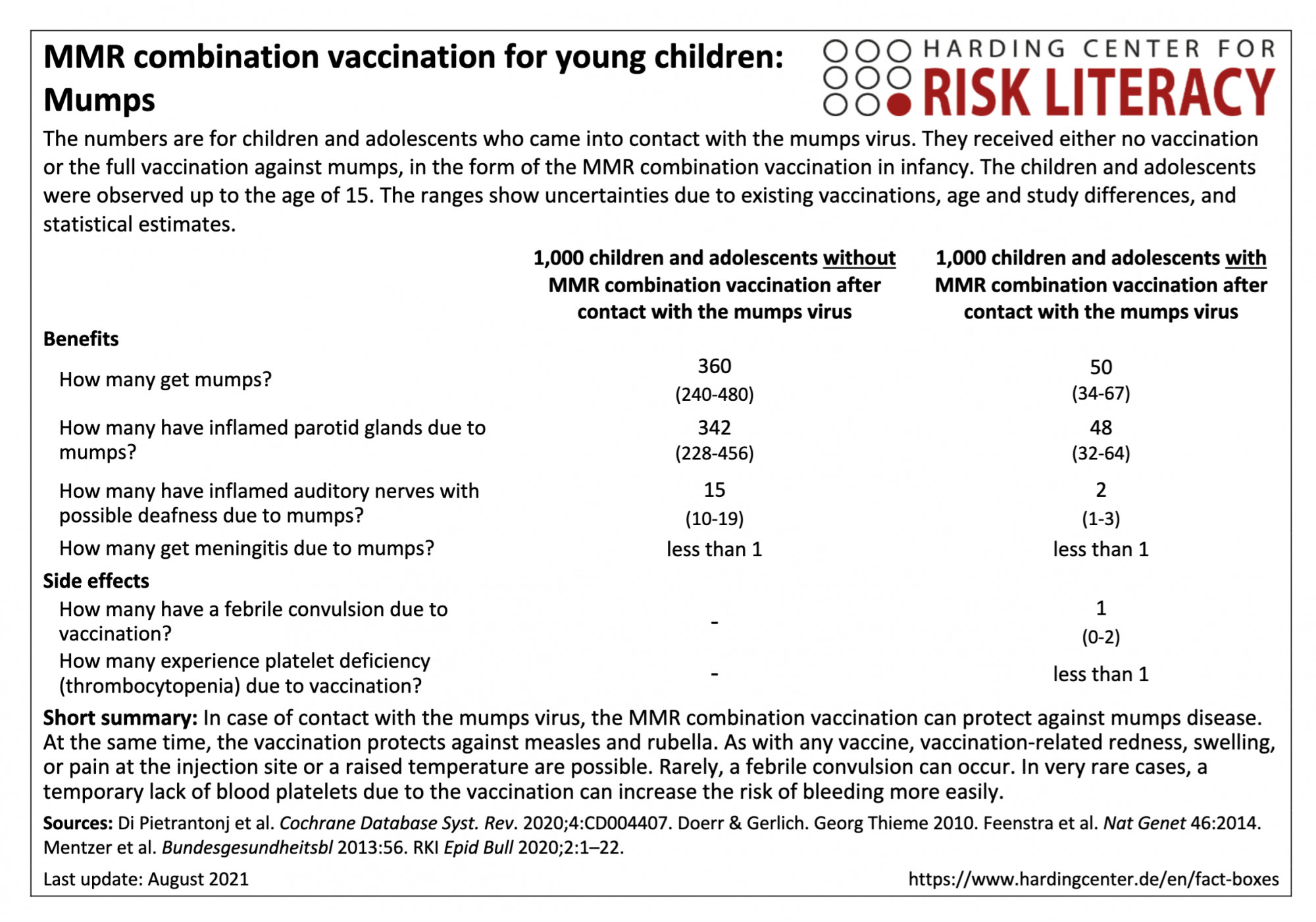
In the fact box, the benefits of the MMR combination vaccination and of the non-vaccination are compared on a model basis for children and adolescents up to the age of 15 who come into contact with the mumps virus. In addition, the side effects of the MMR combination vaccination are compared with those of the non-vaccination.
The table may be read as follows:
If unvaccinated children and adolescents came into contact with the mumps virus by age of 15, 360 (240 to 480) per 1,000 would likely get mumps. In contrast, an average of 50 (34 to 67) per 1,000 of those vaccinated with a combination MMR vaccine would get mumps after contact with the virus [2, 3]. This means that on average 310 out of every 1,000 children and adolescents can be prevented from getting mumps by the MMR combination vaccination.
The MMR combination vaccination can cause febrile seizures in about 1 (0 to 2) out of every 1,000 infants [4].
The figures on the frequency of illness after contact with the mumps virus are based on model calculations with collected data from clinical practice. This includes medical information and textbook data on contact indices (proportion of infected persons among those who have contact with the virus) and manifestation indices (proportion of symptomatically ill persons among those infected). The figures based on clinical practice do not necessarily correspond to the health status of today's population and all vaccines currently offered in Germany [1-3].
The figures on side effects are from various studies with different study designs and populations, with a total of about 2,152,000 participants [4, 5].
| 0-2 Monate | 3 Monate - 5 Jahren | 6 Jahren - 14 Jahren | Ab 15 Jahren | |
| Frauen | - | X | (X) | - |
| Männer | - | X | (X) | - |
Erklärung der Symbole: X = für diese Personen gelten die Zahlen in der Faktenbox; (X) = auf diese Personen lassen sich die Zahlen unter Vorbehalt anwenden (in solchen Fällen ist eine Rücksprache mit ärztlichem Personal empfehlenswert); - = für diese Personen gelten die Zahlen nicht; ? = es ist unbekannt, ob die Zahlen für diese Personen gelten
Because the MMR(V) combination vaccination is a live vaccination with attenuated mumps, measles, and rubella viruses (and chickenpox), mild, non-transmissible "vaccine measles" may occur one to four weeks after vaccination in about 20 to 50 out of every 1,000 infants vaccinated. This is accompanied by fever and a mild measles-like rash. Mild swelling of the parotid glands (swelling of the cheeks) is also occasionally possible. Joint pain has been reported in adolescents and adults (very rarely in children). Rarely, mild swelling of the testicles has been observed. Such vaccination reactions to the MMR(V) combination vaccination are usually temporary and subside on their own without any further consequences [2].
The likelihood of contact with the mumps virus depends on many factors. These include, for example, the number of vaccinated people in the population and the possibilities for the mumps virus to spread. Contacts with the pathogen become less frequent if a majority of the population is vaccinated, which prevents the virus from spreading (herd immunity) [1].
There is no evidence for a connection between the MMR(V) combination vaccination and the occurrence of inflammatory bowel diseases, such as chronic inflammation of the gastrointestinal tract (Crohn's disease) or of the colon (ulcerative colitis). There is also no link between vaccination and the occurrence of autism (profound developmental disorder) or progressive, usually fatal brain damage (subacute sclerosing panencephalitis (SSPE)) [1-2, 5-8].
Furthermore, there is no evidence for a connection between the MMR(V) combination vaccination and cognitive developmental delays, the development of type 1 diabetes, asthma, chronic inflammatory skin diseases (dermatitis/eczema), hay fever, blood cancer (leukaemia), the chronic inflammatory neurological autoimmune disease multiple sclerosis, gait disorders and bacterial or viral infections [5].
The evidence base was determined by the authors of the included review and the creators of the fact box. The overall quality of the evidence is moderate.
The figures on the benefits of MMR(V) combination vaccination were extrapolated using contact and manifestation indices. These have not been explored with randomized controlled trials. However, vaccine effectiveness has been confirmed across different study populations with different study designs. These demonstrate a reduction in disease symptoms in vaccinated versus non-vaccinated individuals.
The findings on seizures due to fever and platelet deficiency could be modified by further research (moderate evidence).
- August 2021 (update of the search, update of the evidence, update of the accompanying text)
- April 2016 (development)
Information for the fact boxes were obtained from the following sources:
[1] Institute for Quality and Efficiency in Health Care (IQWiG). (2020, 2 November). Mumps. gesundheitsinformation.de. https://www.gesundheitsinformation.de/mumps.html (11.08.2021).
[2] Robert Koch Institute (RKI). Communication of the Standing Commission on Vaccination at the Robert Koch Institute: recommendation and scientific rationale for the alignment of occupationally indicated measles-mumps-rubella (MMR) and varicella vaccination. Epid Bull 2020;2:1-22. DOI: 10.25646/6447.3.
[3] Doerr, H. W. & Gerlich, W. H. (2010). Medical virology. Thieme. DOI: 10.1055/b-001-2163.
[4] Feenstra, B., Pasternak, B., Geller, F. et al. Common variants associated with general and MMR vaccine-related febrile seizures. Nat Genet 46, 1274-1282 (2014). DOI: 10.1038/ng.3129.
[5] Di Pietrantonj, C., Rivetti, A., Marchione, P., Debalini, M. G., & Demicheli, V. (2020). Vaccines for measles, mumps, rubella, and varicella in children. Cochrane Database Syst Rev, (4). Art. No.: CD004407. DOI: 10.1002/14651858.CD004407.pub4.
[6] Federal Centre for Health Education (BZgA). (n.d.). Measles vaccination in children. infektionsschutz.de.
https://www.impfen-info.de/impfempfehlungen/fuer-kinder-0-12-jahre/masern/ (12.08.2021).
[7] Mentzer, D., Meyer, H., Keller-Stanislawski, B. Safety and tolerability of monovalent measles and combined measles, mumps, rubella and varicella vaccines. Bundesgesundheitsbl. 2013;56. Available at: https://www.pei.de/SharedDocs/Downloads/wiss-publikationen-volltext/bundesgesundheitsblatt/2013/2013-sicherheit-impfstoffe-masern-mumps-roeteln.pdf (11.08.2021).
